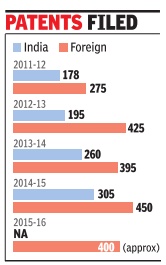
Patents: India
This is a collection of articles archived for the excellence of their content. |
Contents |
India’s patent regime
‘West should learn from India’s high patent standards
SA Aiyar, The Times of India, 7 April 2013
The [Indian] Supreme Court’s denial of a patent for Glivec, an anti-leukaemia drug made by Novartis of Switzerland, has been widely but wrongly hailed by NGOs and castigated by pharmaceutical companies as an attack on patents and a victory for cheap medicine. Actually, the Court fully upheld the principle of patents, but set a high bar for deciding what’s innovative and what’s mere tweaking.
Till 1995, India refused to patent drug molecules. But as a consequence of WTO membership, India in 2005 allowed product patents for drugs, but only for innovations after 1995. This meant no patent for Glivec, which was patented first in 1993. To get around this, in 2006 Novartis tried to patent a new variation of Glivec, for which it claimed improved efficacy. Some other countries granted patents for this variation. But the Indian Patents Office rejected the claim as insufficiently innovative. So too has the Supreme Court.
Many NGOs hailed the judgment for the wrong reason. The Cancer Patients Aid Association, which led the fight against Glivec, declared: “We are happy that the apex court has recognised the right of patients to access affordable medicines over profits for big pharmaceutical companies through patents.”
False: no such right was recognized by the court. It simply said Novartis had not proved that the new variation was innovative enough. The court clarified that it would grant patents for variations that were more efficacious, but set a higher standard for proof than many western courts do.
NGOs are wrong to paint Novartis as a bloodsucker that pauperizes patients. Glivec has saved lakhs of leukaemia patients from death. This is a great boon, and we must encourage more such life-saving boons by granting patents for new drugs. However, this does not mean giving patents for mere tweaking and “evergreening” of existing drugs through minor variations.
Patents are supposed to spur innovation, but when granted over-liberally they create so much lawsuit risk and cost that they end up hampering innovation, not aiding it. The Economist (UK), no basher of multinationals, acknowledges that over-liberal proliferation of patents fuels many of the American patent system’s broader problems, such as patent trolls (speculative lawsuits by patentholders who have no intention of actually making anything); defensive patenting (acquiring patents mainly to pre-empt the risk of litigation, which raises business costs); and “innovation gridlock” (the difficulty of combining multiple technologies to create a single new product because too many small patents are spread among too many players).”
Patent trolls buy up patents in bulk, typically from bankrupt companies, not for actual use but simply to hit other innovators with lawsuits for patent infringement, forcing them to settle to avoid fat legal bills. One US study estimated such legal costs at $ 29 billion in 2011 alone.
Last year, Google bought Motorola’s failing smartphone business for $ 12.5 billion, to access its 17,000 patents. Microsoft and others paid $ 4.5 billion for 6,000 patents from Nortel. Most of these will never be used in actual production: they are simply kept as legal weapons for possible lawsuits. This has nothing to do with innovation, which patents are supposed to promote.
The West’s over-liberal patent system is broken. It should learn from India’s much tougher system. Patents should be seen as monopolies, to be given sparingly only for genuine innovations where the public benefit clearly exceeds the monopoly cost. This means setting a high bar for innovation. High standards are desirable for patents, as for everything else.
CSIR guidelines: filing of Indian, foreign patents/ 2016
Akshaya Mukul, CSIR wants labs to stop random filing of patents, Oct 19 2016 : The Times of India
The Council of Scientific & Industrial Research (CSIR) has sent out a stern message to its 38 laboratories to avoid indiscriminate filing of Indian and foreign patents.
In order to “inculcate responsible behaviour“ CSIR has decided that “henceforth 25% of expenses incurred on prosecution and maintenance of Indian patents and up to 50% of expenses on foreign patents will be borne by the laboratories“. CSIR director-general Girish Sahni has told laboratories “if they succeed in licensing, headquarter will give you a matching grant“.
In a strongly worded letter to lab heads, Sahni said “patents are just filed for the sake of filing without any techno-commerical and legal evaluation“. He told them that even choice of countries where patents are being filed is “ad-hoc without any logic“. For instance, he said, majority of patents are “bio-data“ patents.
“Individual scientists are using them for getting promotions and labs are pla ying number game. Once the patent is granted, neither the scientist nor the lab bothers about it. There is no serious attempt to find licensee and a review system to periodically look at the portfolio does not exist,“ he said.
In this regard, Sahni pointed out, “Renewal is simply done at the recommendation of the scientist, as a result some patents are maintained till scientist retires. After that labs have no documentation available to even demonstrate the know-how.“
He added that patents are being abandoned after filing and are being allowed to lapse in early years. This, he said, shows that “no due diligence was done before filing but in the process we are spending filing fees and paying attorney fees running into crores of rupees“.
To add to it, he said, there is a cost of manpower in the laboratories for applying to International Patent Union.
“All these expenses goes down the drain. Please note that each Indian patents costs us Rs 2 lakh and cost of US patent is $20,000-plus,“ he said.
Sahni gave the instance of 2015-16 when nearly 400 foreign patents were filed. “If each patent is taken to logical conclusion, our total expenditure will work out to be Rs 56 crore. A poor nation cannot afford to spend this kind of money every year to sustain our ego of being intellectual capital of India,“ the director-general said.
He has told each lab director to take “personal responsibility for IPR and business development, and must put in place systems and processes so that worthless patents are not filed and demonetisation is pursued vigorously“.
Amended Patent Rules reduce processing time
Patent offices in India, traditionally known to be long-winding and tardy , are witnessing a major overhaul.For the first time ever, a patent has been granted within a record 113 days. Usually , the process takes five to six years.
Using a new provision under the amended Patent Rules, Hyderabad-based Optimus Drugs and more than 15 other companies -including startups and research-based firms -have been granted patents within a record time of 113-300 days, official sources told TOI.
They added the move will spur innovation and bring the patent office in line with global practices. The provision -`expedited examination of patent applications' -aims to shorten the time period for grant of patents from the `filing' of application to its `publication' to two-three years, down from the current five-seven years.
Under the amendment in Patent Rules (2003) on May 16, 2016, India has introduced `expedited examination' for patents filed by startups, and those entities which select India as the competent Internatio nal Search Authority (or ISAs, with 20 such authorities globally). Optimus Drugs was granted a patent on Apixaban within the shortest time, among others filed under the new provision, according to the controller general of patents, designs & trade marks, OP Gupta.
He added the rules aim to remove bottlenecks in the process, clear pendency and encourage more filings, technological advancement and innovation. The modified rules will allow future applicants to put applications on fast track if they select India as an ISA, or International Preliminary Examining Authority , and file applications in India first.
Intellectual Property Organization: a new treaty: 2024
May 25, 2024: The Times of India
New Delhi : Members of the World Intellectual Property Organization (WIPO) have finalised a new treaty that will make it mandatory for patent applicants to disclose the country of origin or the source of genetic resources if the claimed invention is based on those materials or associated traditional knowledge.
The treaty, that will provide additional protection for Indian genetic resources and traditional knowledge, such as yoga, siddhi and ayurveda, has been a key demand of the developing world, led by India.
The Geneva-based agency, responsible for evolving rules related to patents and trademarks, finalised the agreement Friday, which will require several countries to re- work their domestic regulations. India has insisted on global giants giving credit to the vast traditional knowledge and genetic resources instead of seeking to exploit them by registering patents.
While protection against misappropriation is available in India, countries, which do not have disclosure of obligations, the disclosure requirements will provide for protection across countries. At present, 35 countries have some form of disclosure obligations, most of which aren’t mandatory and don’t have appropriate sanctions in place for effective implementation.
The WIPO treaty on intellectual property, genetic resources and associated traditional knowledge, will for the first time take note of the knowledge and wisdom which have supported economies, societies and cultures for centuries and are now inscribed into the global IP system. “These are historic achievements championed by India as a provider of traditional knowledge and wisdom and repository of biodiversity. s a significant win for countries of the global South and for India,” said a govt official.
The resistance from countries such as the US and multinationals is evident from the fact that it took 150 countries to evolve a consensus after two decades of negotiations.
Trade Mark Rules
‘Well-known trademarks’
Lubna Kably, India Inc can now register trademark as `well-known', March 8, 2017: The Times of India
Only Courts Had Power To Take Decision Earlier In Disputes
In a significant development, the Trade Mark Rules 2017 -which were notified on March 6 -permit companies to apply for their trademark to be recognised as a `wellknown' one. In simple terms, a well-known trademark is one that is well recognised and any infringement could result in misleading the public.
Prior to this notification, any trademark was held as `well-known' by courts only consequent to a dispute regarding its use, such as when another party applied for registration of the same or similar trademark. IPR advocates have many examples to share.Amul' for instance, was given ` the status of a `well-known' mark, owing to infringement of the mark in the name of `IMUL' by another milk cooperative. Watch manufacturer `Rolex' got a favourable order from the Delhi high court, which recognised `Rolex' as a well-known mark, and restrained Alex Jewellery from using this name for artificial jewellery as it could be misleading the public. Other wellknown decisions are in the case of Whirlpool, DaimlerBenz, to name a few.
“While many Indian com panies who have already registered their trademarks will continue to get protection against registration of conflicting trademarks, the rules will be a boon to foreign companies that do not have a registered trademark in India but at the same time wish to protect themselves. At times, entry of some companies may be restricted -say, an adult ma gazine. Yet, through the process of registration of wellknown trademarks, it will now be easier for the publication to protect its globally recognised trade name. This should also reduce the cost of opposition as, typically , the registrar will cite the registered well-known trademarks as conflicting marks to subsequent applications,“ explains Gowree Gokhale, partner and head of IPR practice at Nishith Desai Associates.
Raja Selvam, managing attorney at Chennai-based law firm Selvam and Selvam, says, “Recognition of a trademark as a well-known trademark will greatly help the trademark owner in opposition or infringement proceedings (eg: Amul vs Imul).The registration fee of Rs 1 lakh is insignificant if compared to the savings in potential litigation costs.“
Intellectual property, India: 2006-15
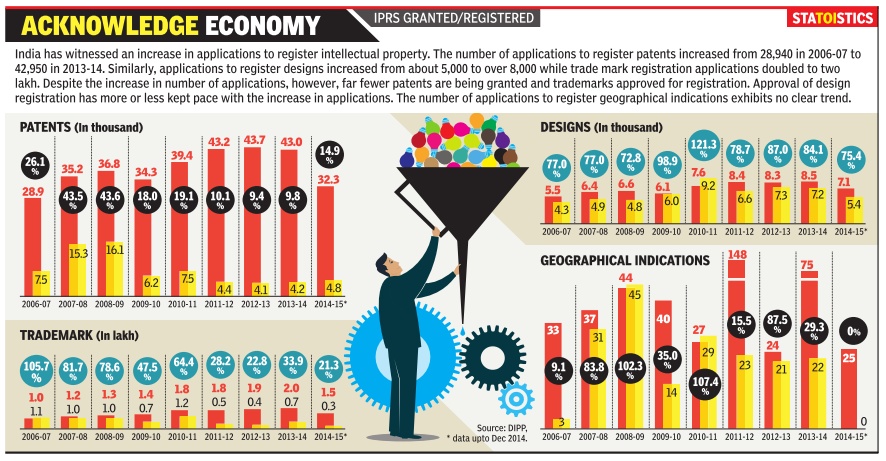
See graphic:
2006-15, the registration of patents, trademarks, geographical indications and designs in India, applications and approvals
Expedited patents
2016-18
Faster patents for women, small biz, December 26, 2018: The Times of India
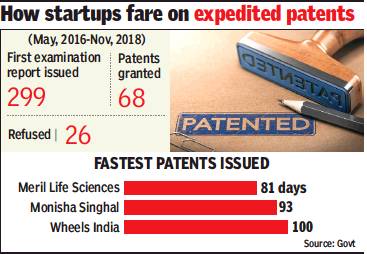
From: Faster patents for women, small biz, December 26, 2018: The Times of India
The government has proposed to fast-track patent applications from women and small businesses in its bid to encourage innovation from new category of innovators.
Currently, the benefit of expedited patents is available to startups, with over 350 companies taking advantage of the special window that was opened in May 2016 as part of the government’s flagship Start-up India scheme. Now, the department of industrial policy and promotion (DIPP), has floated draft rules to amend the Patent Rules, 2003 and extend the facility to other groups, including Indian women as part of a policy to encourage innovation and entrepreneurship. India is probably the only country to look at encouraging patent applications from women, a government official said.
The draft rules, released earlier this month, have also suggested that the window be opened to government undertaking. While no time frame has been proposed, the fastest patent application under the expedited window was granted in 81 days, data accessed by TOI showed.
Since the special facility was given to startups, close to 1,275 applications have been filed, of which 646 were filed from Chennai. Mumbai was at number 2 with 321, and Delhi was third with 268 applications. Of these, 350 applications were for expedited clearance.
In recent years, the government has sought to reduce the time taken for processing patent applications by creating more capacity in the Patents Office, besides roping in professionals from IITs to help process the proposals. Against the average time of five-seven years taken in clearing patent applications earlier, the government is looking to cut down the time to 18 months by March 2020.
the Indian Patent Office granted 13,045 patents in 2017-18, compared to 9,847 in the previous year with a bulk of the applications processed being filed by overseas companies and individuals.
2015-19: Expedited patents for all
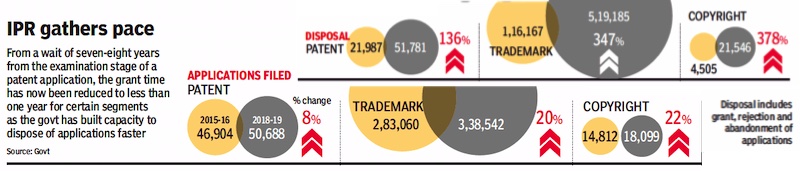
From: July 14, 2019: The Times of India
See graphic:
2015-19: Reduction in the grant time for patents
India and the world
2022
Rupali Mukherjee, Nov 7, 2023: The Times of India
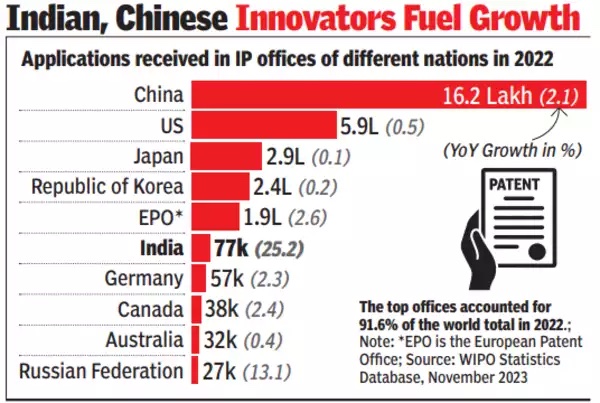
From: Rupali Mukherjee, Nov 7, 2023: The Times of India
NEW DELHI: Global patenting activity soared to new records in 2022, fuelled by Indian and Chinese innovators and powered by increased levels of innovation, entrepreneurship and digitalisation in these countries. Patent filings by Indian applicants grew by 31.6% in 2022, extending an 11-year run of growth unmatched by any other country among the top 10 filers, according to WIPO's (World Intellectual Property Organisation) annual World Intellectual Property Indicators (WIPI) report.
What is even more significant is that resident patent filings in India (filed with the Indian Patent Office) rose by 47% to 38,551 in 2022, and were half of the total filings of 77,068 filed with the Indian Patent Office. So, for the first time ever, there were more patents filed by Indian residents as against foreign filers, reflecting India's fast growing innovation ecosystem, data culled from WIPO said.
Innovators from around the world submitted 3.5 million patent applications in 2022, marking a third consecutive year of growth. Overall, China, the US, Japan, Republic of Korea and Germany were the countries, with the highest numbers of patent filings in 2022.
"IP filings have braved the pandemic to continue to grow, powered by increased levels of innovation, creativity, entrepreneurship and digitalisation in all parts of the world. Developing countries are increasingly becoming engines of IP, showing the greatest growth rates as they harness the innovation and creative potential of their people. However, uncertainty continues to weigh on the global innovation ecosystem, with venture capital funding dropping in many parts of the world. We urge investors to pursue quality, but not at the expense of supporting good ideas that can change the world for the better, WIPO DG Daren Tang said.
Continuing a longer-term trend, bulk of IP filing activity occurs in Asia, from all origins. Asia accounted for 67.9%, 67.8% and 70.3% of global patent, trademarks and industrial designs filing activity in 2022. The largest rise in filings was reported from India. Switzerland (6.1%), China (3.1%), Austria (2.5%) and the UK (2.5%) also reported robust growth in filings.
Patents applied for by India
2016-17
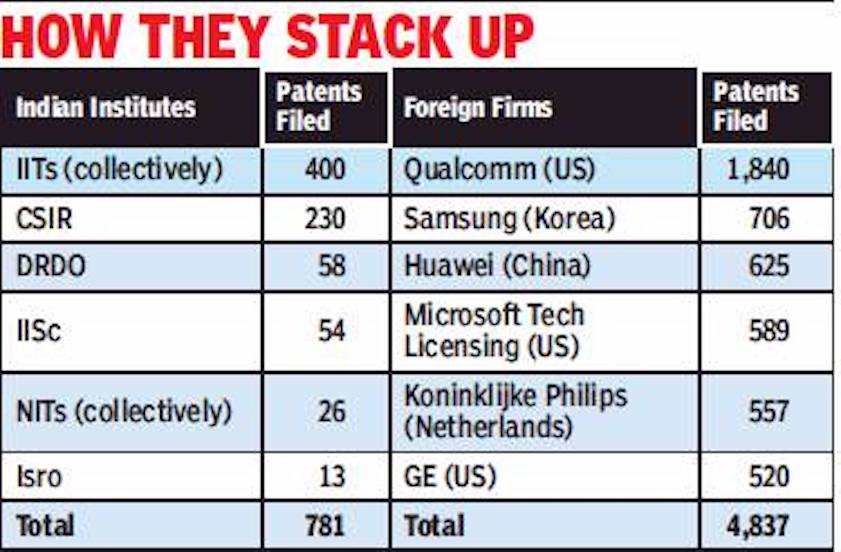
From: Chethan Kumar, 1 US co files 135% more patents in India than do all top desi labs, July 24, 2018: The Times of India
The number of patents filed by a US firm in India in 2016-17 was more than double of all patent applications from 50 labs under DRDO, more than 40 labs in IISc, 38 labs under CSIR, 31 NITs, 23 IITs and six major research facilities under Isro.
While all these institutes together filed 781 patent applications, Qualcomm Inc — a US semiconductor and telecom equipment firm — filed 1,840 applications, according to the Controller General of Patents, Design and Trademark.
India, which lags behind other countries whose patents outnumber India’s by huge margins globally, suffers the same problem even domestically, with foreign countries filing way more patents than locals. In 2016-17, 45,444 patents were filed in India, of which 71% were filed by foreigners (inventors and applicants) and other foreign applicants (legal entities), while 29% were filed by Indians. The US, Japan and China together account for more patents filed in India than the entire Indian scientific, research and business communities.
Only 2.9 of every 10 patents filed in India are by Indians
In a continuing trend of foreign domination, only 2.9 of every 10 patents filed in India as per data for 2016-17 are by Indians, a marginal improvement from 2.8 of every 10 patents in 2015-16. The information for 2017-18 will be released later this year.
Renowned scientist Prof Roddam Narasimha says, “We don’t lack talent or entrepreneurship but we lack the ecosystem. Work in science and innovation is abysmal because there’s been a legacy of poor policy and low confidence to take risks. But that has been changing over the past few years.”
Of all foreigners filing patents, 10 big companies account for 19% and data shows that Qualcomm Incorporated continues to top the list, followed by Samsung Electronics and Huawei Technologies. A country wise break-up of patents filed shows that the US, Japan and China, which occupy the top three positions, together account for 14,636, which is 1,659 more than patents by Indians.
Of the 45,444, just 10 countries filed 22,970, including countries like Netherlands (1,295), and experts say India has a long way to go before the scenario changes, even as they point out that most foreigners only file patents in India to prevent reproduction of their technology.
“We need to bring in the patent culture, which is not there in all institutes. We have a handful of people whose innovations are good enough for patents, and that needs to change,” said K Kasturirangan, former Isro chairman and head of the committee formulating India’s new education policy. In the period between 2013-14 and 2016-17, 1.9 lakh patent applications were filed in India, of which 1.37 lakh (72%) were filed by foreigners.
Patent success rate: 2010-19
Chethan Kumar, March 1, 2021: The Times of India
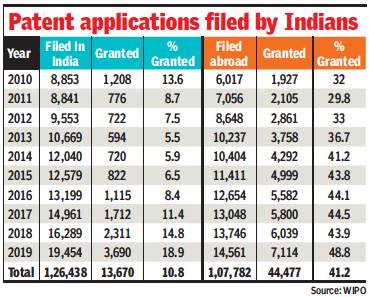
From: Chethan Kumar, March 1, 2021: The Times of India
Indian researchers, scientists and innovators appear to have more success in obtaining patents when they file applications in another country. Analysis of data for 10 years shows that on an average, four out of every 10 applications filed by Indians abroad fetch a patent, while it is one-in-10 in India.
From slow disposal to indiscriminate filing and low cost of registering patents, experts and researchers point to a variety of reasons for this trend.
According to data from the World Intellectual Property Organisation, Indians filed 1.2 lakh applications in the country between 2010 and 2019 and 13,670 were granted patents. Comparatively, 44,477 of the 1.07 lakh applications filed by Indians abroad were granted. “The process is really timeconsuming in India. It takes four to five years,” Sourabh Gargav of JNCASR said.
3.2L applications filed by foreigners in India in 10 years, 76k got patents
Muniasamy Neerathilingam, a translational researcher, echoing Gargav’s views said another reason could be that those filing abroad are more serious claims. An official from the Indian patent office said a lot of applications filed in India are to pip competition and obtain first mover’s advantage, and that researchers take time to strengthen their claims.
Rajendra Ratnoo, controller general of patents, designs & trademarks, told TOI: “In the last five years, the disposal rate has increased. While we encourage patent applications, we also have a robust system that’s meticulous in selection, which may be resulting in many applications not getting converted into patents. Given the cost of filing abroad, applicants are careful and file only such claims they know have a chance of getting cleared.”
Another official said the cost of the entire official filing fee is less than Rs 10,000 compared to $2,000 (around Rs 1.5 lakh) in the US. “...There are other costs like attorney fees, which are also affordable in India but the cheapest lawyers in other countries are expensive. Applicants are mindful of making claims, which is why more applications are cleared abroad,” he said. Year-on-year data shows an increase in clearance in the last five years. For example, just 8.7% and 5.9% of the applications were granted patents in 2011 and 2014, respectively, the same jumped to 14.8% and 18.9% in 2018 and 2019. With 43.9% and 48.5% of the applications filed abroad getting grants in 2018 and 2019, the gap in the success rate continues to persist.
Foreigners continue to file more applications in India compared to domestic researchers. This data shows that foreign patent filers have a better chance of getting a patent in India with 23.4% of all applications getting approved compared to just 10.8% filed by Indians. Foreigners filed over 3.2 lakh applications in India in these 10 years, of which 76,637 were granted patents.
Patents filed in India: 2010> 2021
February 1, 2022: The Times of India
Chennai: With intellectual property being the backbone of a knowledge-based economy, the Economic Survey 2021-22 notes that India is on the right path with filing and granting of patents registering a steady rise and more Indians innovating and applying for patents.
The number of patents filed in India increased by almost 50% from 2010-11 to 202021, the Economic Survey showed. Number of patents filed stood at 39,400 in 2010-11, and 58,502 in 2020-21.
TNN
2017- 22
January 31, 2023: The Times of India
NEW DELHI: Patent filings by Indian applications were estimated at 10,918 during the December quarter, compared with 10,340 applications lodged by non-domestic entities, marking the second time during 2022 when quarterly filings by domestic players were higher.
Data available with the department for promotion of industry and internal trade showed that during January-March 2022, of the 19,796 patent applications filed, 10706 were filed by Indian applicants, against 9090 by non-Indian applicants. On Sunday, in his monthly radio broadcast, ‘Mann ki Baat’, PM Narendra Modi had referred to this number to point out how for the first time in 11 years filings by Indian players exceeded those from outside the country.
During the current financial year, the first half (April-September), however, saw a reversal of the trend with non-Indian patent applications beating those by domestic entities. As a result, during the nine months ended December 2022, of the 57,783 applications, close to 30,000 are by non-Indian players.
Trends available so far, however, indicate that the applications filed in 2022-23 will exceed last year’s number of 66,440.
Over the last few years, the government has been laying emphasis on patents and has sought to streamline the system for filing applications, their examination and grant. As a result, there has been a 39% jump in the number of applications filed between 2017-19 (47,854) and last year (66,440). The patents granted rose at a much sharper pace of 131% to 30,074 last year, compared to 13,045 in 2017-18.
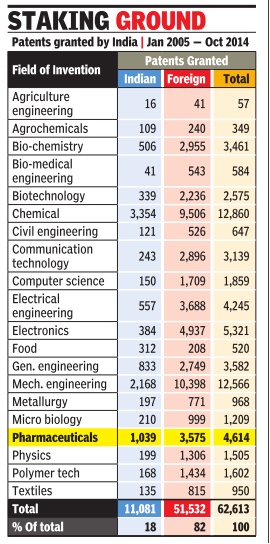
Patents granted by India
In 2005- 14
Jan 27 2015, Sushmi Dey
Promote Indian drugs, US health groups urge Obama
Leading US health groups including AVAC, Oxfam America, amfAR, Health Global Access Project (GAP), TAG (Treatment Action Group)and others have written to Barack Obama urging him to support India in providing “high-quality, low-cost generic medicines essential for health care around the world“. This comes in the wake of the two nations agreeing to enhance engagement on IPR at the latest bilateral talks. The joint statement after the Obama Modi meeting said the two countries expressed interest in sharing information and best practices on IPR, and reaffirmed commitment to stakeholders' consultations on related policy matters.
There are concerns that US is pressuring India to push for liberal policies in favour of pharma MNCs seeking patent protection for their products in India.
“Instead of using your trip to promote the narrow interests of one segment of the pharmaceutical industry, we ask you to support the interests of people who need affordable medicines, whether they live in the US, in India, in Africa or elsewhere,“ civil society organizations said in the letter.
In April, the US brought out the Special 301 report taking unilateral measure to pressure countries to accept IPR protection beyond WTO obligations. The report classified India as a `priority watch list country' , highlighting concerns related to Indian patent law, grant of compulsory licence and inclusion of a statement relating to CL for green technologies in India's manufacturing policy .
Adding Indian laws are compliant to the WTO TRIPS agreement, the civil society letter said, “Recent US policy stances have sought to topple parts of India's intellectual property regime that protect public health to advance interests of multinational pharmaceutical corporations in longer, stronger, and broader exclusive patent and related monopoly rights.“
India is a major manufacturer and exporter of low cost generic medicines. The domestic pharmaceutical industry registered exports worth $14.84 billion in the 2013-14. While the US itself contributes the majority of this sales, India is also a major supplier of generic medicines to other parts of the world.
While India is currently in the process of finalizing an IPR policy and a separate policy for pricing of patented medicines in the country, bilateral talks between the two nations along with Obama's visit to India assume significance not only for the Indian pharma industry but for patients across the world.
2005-18
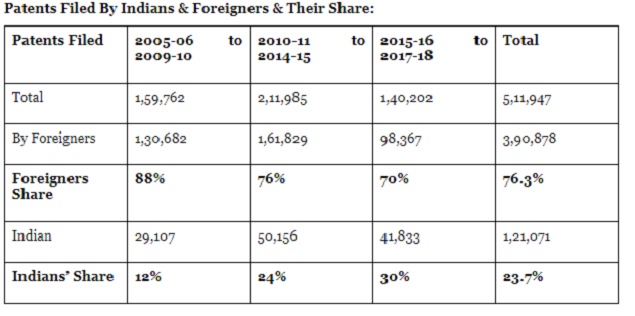
From: Chethan Kumar, 76% of patents filed in India in last 13 years were by foreign companies, September 10, 2020: The Times of India

From: Chethan Kumar, 76% of patents filed in India in last 13 years were by foreign companies, September 10, 2020: The Times of India
BENGALURU: Nearly eight out of every ten patent applications filed in India in more than a decade have been by foreigners residing abroad. A majority are by foreign firms, even as the number of Indian applications is slowly increasing.
More than 5.1 lakh patent applications were filed in the country between 2005-06 and 2017-18 according to the data compiled by the Department of Science and Technology (DST) as part of the latest Science & Technology Indicators (STI 2019-20) released last month.
And, an analysis of the data shows that more than 76% of these applications were filed by "foreigners resident abroad", as the government classifies them and the rest were by "Indians."
This is a trend experts say India must arrest. “We have to be more innovative,” Biocon Ltd executive chairperson Kiran Mazumdar Shaw said.
A senior defence scientist told TOI that India does not lack talent and that even investments are improving, especially in the private sector. He said India lacked the right ecosystem. The TOI had reported last month about how R&D investments by the private sector has been growing at twice the rate of public/government investment.
In March 2016, noted scientist Prof CNR Rao, who has consistently batted for increased funding for science and had remarked: “Researchers are relaxing with too much furniture!”
Among the patents filed by foreigners, most are individuals or companies from the US, Japan and Germany, accounting for more than 45% of the patents in the said period, while the UK, France, Switzerland, Russia, Netherlands and Italy also had hundreds of applications filed.
With nearly 1.4 lakh applications, patents filed by those from the US alone accounted for more than 27% of total applications filed in the said period.
Former Isro chairman K Kasturirangan, who also headed India’s committee on new education policy told TOI: “...We need to really improve research content at major institutions and particularly at the 900-odd universities in the country. We have to make sure that pure research becomes part of applied research, which goes into translational and industrial research. Unless this four-pronged attack is made on research to create more innovation and ideas, the question of patent doesn’t arise.”
In fact, the proposal for the National Research Foundation (NRF) in the education policy, he said, is the reflection of concerns about some of these outcomes of research. “I hope that we improve research as fast as we can and create an environment that leads to an ecosystem of more innovation and products that are patentable,” he added.
Qualcomm, Huawei Top Foreign Cos
The STI-2019-20 also provides data on top foreign companies filing patents in the five years between 2013-14 and 2017-18. Analysis shows that most of the top 19 companies were tech and telecom firms, including Chinese MNC Huawei Technologies.
Qualcomm Incorporated—an American semiconductor and telecommunications equipment firm — tops the table with 6,960 applications, followed by Dutch multinational firm Koninklijke Philips, which had 3,670 applications.
The TOI had reported in July 2018 on how Qualcomm alone had filed more than twice the number of patents than the collective number of applications from 50 labs under the DRDO, 38 labs under CSIR, 23 IITs, 31 NITs, more than 40 labs in the IISc and six major research facilities under Isro, in 2016-17.
Aside from these two firms, Huawei had filed 1,393 patents in the said period, while other big firms like Google, Microsoft Technology Licencing, Siemens, Robert Bosch and others also filed hundreds of applications.
City-wise, 2020-2023

From: June 27, 2025: The Times of India
See graphic:
Patents issued in India. City-wise, 2020-2023
Patents applied for by India, others
2017
January 31, 2019: The Times of India

From: January 31, 2019: The Times of India
See graphic:
Patents applied for by Indians and others and patents granted, in 2017
Worldwide patent applications reached a whopping 3.17 million in 2017. This is a 5.2% increase over the previous year. Interestingly, it is China patent offices that received the most number of applications, followed by the US and Japan. Asian patent offices received 65% of all applications filed worldwide. India received 46, 582 applications.
2018: India Leads In Top 15 With 27% Growth

From: Rupali Mukherjee, In a 1st, Asia generates over half of patents filed in 2018, March 27, 2019: The Times of India
India Leads In Top 15 With 27% Growth
Asia-based innovators filed over 50% of all international patent applications for the first time in 2018. The development came on the back of significant growth from India, China and Republic of Korea (South Korea), according to data from the global intellectual property (IP) services of the World Intellectual Property Organization (WIPO).
India filed a total of 2,013 international patent applications, registering the highest growth of 27% among the top 15 countries. India and China are the only two middle-income countries among the top 15 origins of Patent Cooperation Treaty (PCT) applications. Though India showed huge growth, its share in global filings was a minuscule 0.8%, while China was ranked second with a huge 21% share.
Top patent filers from India are led by Chennai-based TVS Motor Company. Institutions like Council of Scientific and Industrial Research, and Indian Institute of Technology are ranked second and third, followed by Reliance Industries, and educational institution Indian Institute of Science. But the list is dominated by pharmaceutical companies including Dr Reddy’s, Cipla, Lupin and Sun Pharma. Mylan, Hetero, MSN Labs and Wockhardt are the other pharma companies that made it to the list of top filers.
Overall, WIPO’s PCT passed a record-breaking quarter million (253,000) filings in 2018 — a nearly 4% increase. USbased applicants filed 56,142 PCT applications, followed by China (53,345) and Japan (49,702). Germany and South Korea ranked fourth and fifth, respectively, with 19,883 and 17,014 filings. Over half of all PCT applications came from Asia (50.5%), with Europe (24.5%) and North America (23%) accounting for about a quarter each.
“Asia is now the majority filer of international patent applications, which is an important milestone for that economically dynamic region and underscores the historical geographical shift of innovative activity from West to East,” said WIPO director general Francis Gurry.
Among fields of technology, digital communication (8.6%) overtook computer technology (8%) with the largest share of published PCT applications, followed by electrical machinery (7%), medical technology (6.7%) and transport (4.6%). Among the top 10 technologies, transport (11%), digital communication (10%) and semiconductors (9.8%) showed the highest rates of growth during the year.
Globally, the top 10 applicant list comprises six companies from Asia, two each from Europe and the US. China-based telecom giant Huawei Technologies, with a record number of 5,405 PCT applications, was the top corporate filer in 2018. Japan-based Mitsubishi Electric ranked second (2,812 filings), followed by Intel (2,499) and Qualcomm (2,404), both from US, and ZTE of China (2,080).
Among educational institutions, University of California was the largest user of the PCT system with 501 applications, extending a leading position it has held since 1993. Massachusetts Institute of Technology (216) ranked second, followed by Shenzhen University (201), South China University of Technology (170) and Harvard University (169).